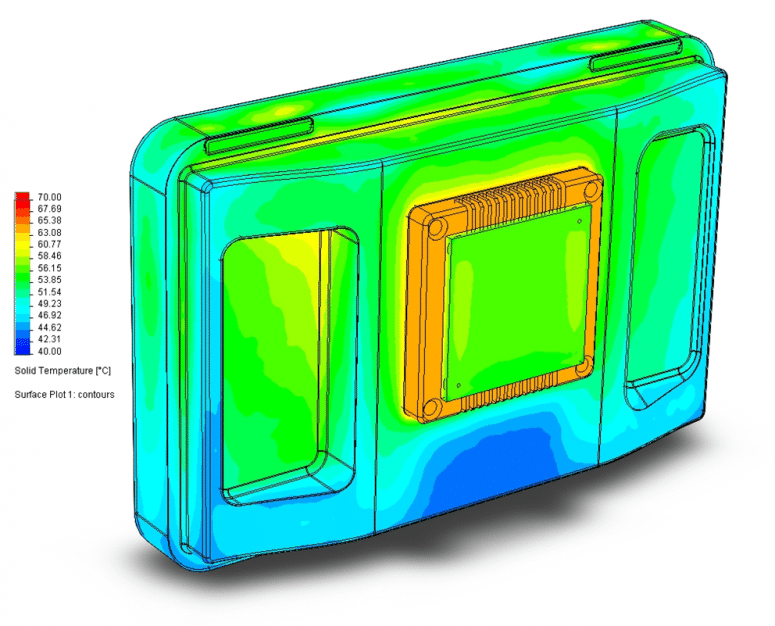
COM and carrier board in next generation medical instrument

Hectronic H6049
• Intel® Atom™ Z510/Z530 CPUs
• Intel® Atom™ Z510/Z530 CPUs
• Processor speeds: 1.1GHz, 1.6GHz
• Extended/Industrial Temperature, optional
• Soldered DDRII DRAM
• Onboard NANDrive Flash SSD
• Trusted Platform Module, TPM 1.2
• LVDS, SDVO
• 7 x USB 2.0 Ports
• Conformal coating, optional